Soft Molecular and Supramolecular Materials
| Soft Molecular and Supramolecular Materials | |
In chemistry the term "soft materials" is used to emphasize weak bonding between molecular species. In soft materials the molecules are held together by non-valent interactions: van der Waals forces, hydrogen bonds, weak coordination and so on. Such materials can instantly form through the process of self-assembly (aka "the bottom-up approach" in nanochemistry) once conditions are suitable and easily transform into various forms when these external conditions change. In spite of weak bonding, the molecules usually build a highly organized, often sophisticated, structure. Soft supramolecular materials display an interplay of molecules of two or more different types combined in a complex but regular architecture. Well-known examples of soft materials are molecular and liquid crystals, many types of inclusion compounds, co-crystals, dendrimers, fibrils and fibers, and gels. |
|
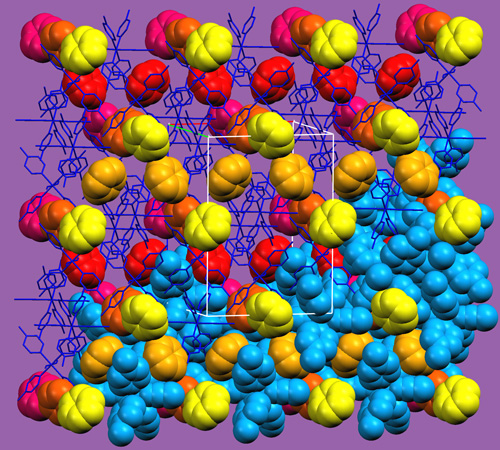
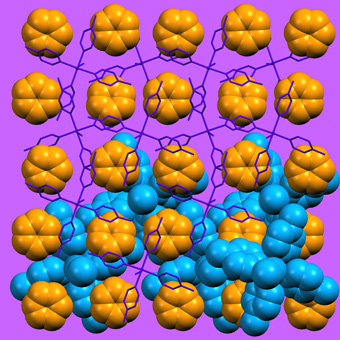
Supramolecular organization in two different inclusion compounds formed by the same host-guest pair (host molecules shown in blue). |
|